Features of Hydride Synthesis
Main Article Content
Abstract
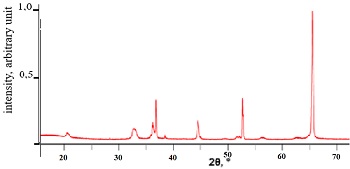
Lithium, sodium, and titanium hydrides have been synthesized. It has been found that lithium hydride is formed at a temperature of 650-700°C and a pressure of 12 atm. Lithium hydride contains 11 mass.% of hydrogen. Hydrogen desorption from lithium hydride occurs at a temperature of 700°C. Lithium hydride decomposes in water to form lithium hydroxide and hydrogen. Sodium hydride is formed at a temperature of 300-450°C with a hydrogen absorption level of up to 4 mass%. Hydrogen desorption from sodium hydride occurs at a temperature of 350-400°C. Titanium hydride is formed at a temperature of 600-650°C with a hydrogen absorption level of up to 4 mass%. Hydrogen desorption from sodium hydride occurs at a temperature of 450-650°C. Lithium, sodium, and titanium hydrides can be used in hydrogen storage and transportation processes.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
- Antonova MM. Properties of Hydrides. Handbook. Moscow: Metallurgy, 1975.
- Mikheeva VI. Transition Metal Hydrides. Moscow: USSR Academy of Sciences, 1960.
- Mueller WM, Blackledge JP, Libowitz GG. Metal Hydrides. Academic Press, New York, 1968.
- Payzullakhanov MS, Parpiev OR, Avezova NR, et al. Hydrogen Storage in Porous Ceramic Materials of Aluminosilicate Composition. Applied Solar Energy. 2022, 58(5): 722-724. https://doi.org/10.3103/s0003701x22601338
- Paizullakhanov MS, Parpiev OR, Shermatov ZhZ, et al. Hydrogen Absorbers Based on Porous Ceramics Synthesized in a Solar Furnace. High Temperature Material Processes An International Quarterly of High-Technology Plasma Processes. 2024, 28(4): 41-46. https://doi.org/10.1615/hightempmatproc.2024053882
- Bhosle V, Baburaj EG, Miranova M, et al. Thermal stability and decomposition of TiH2 in different atmospheres. Materials Science and Engineering: A. 2003, 356(1–2): 190–196. https://doi.org/10.1016/S0921-5093(03)00210-7
- Kovalev DYu, Prokudina VK, Ratnikov VI, et al. Thermal decomposition of TiH2: A TRXRD study. International Journal of Self-Propagating High-Temperature Synthesis. 2010, 19(4): 253-257. https://doi.org/10.3103/s1061386210040047
- Schlapbach L, Züttel A. Hydrogen-storage materials for mobile applications. Nature. 2001, 414(6861): 353-358. https://doi.org/10.1038/35104634
- Bogdanović B, Schwickardi M. Ti-doped alkali metal aluminium hydrides as potential novel reversible hydrogen storage materials. Journal of Alloys and Compounds. 1997, 253-254: 1-9. https://doi.org/10.1016/s0925-8388(96)03049-6
- Ley MB, Jepsen LH, Lee YS, et al. Complex hydrides for hydrogen storage – new perspectives. Materials Today. 2014, 17(3): 122-128. https://doi.org/10.1016/j.mattod.2014.02.013
- Varin RA, Czujko T, Wronski ZS. Nanostructured Hydrides for Solid State Hydrogen Storage for Vehicular Applications. Green Energy. Published online 2011: 223-286. https://doi.org/10.1007/978-1-84882-647-2_6